AMSBIO has launched an enhanced GMP-compliant version of its animal-origin-free, chemically defined StemFit™ iPSC expansion medium.
StemFit™ Basic04 CT is designed to improve the growth and pluripotency of induced pluripotent stem cells (iPSC) and meet the Good Manufacturing Practice (GMP) standards for Active Pharmaceutical Ingredients (API).
Offering superior iPSC performance, expansion, and maintenance of long-term genetic stability – StemFit™ Basic04 CT is optimized for single cell passaging, making it ideal for use in large-scale production of stem cells.

Bottle of StemFit Basic04 CT
The manufacturing and quality control of this exciting new product are managed in strict accordance with GMP standards. All bottles of StemFit™ Basic04 CT come provided with a certificate of analysis and any documentation required for a seamless transition to clinical applications.
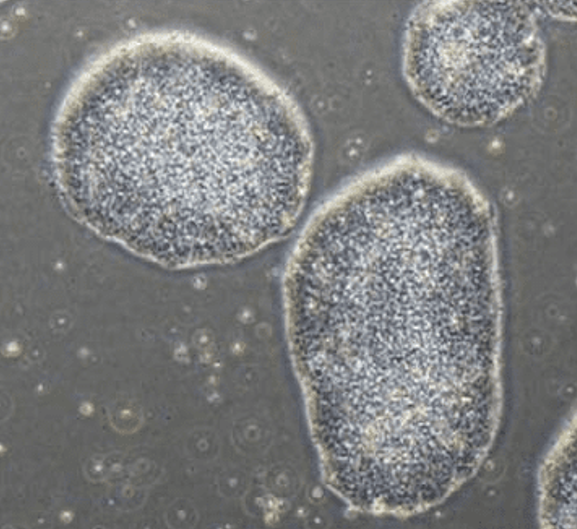
Colony of iPSCs grown using StemFit Basic04 CT
Aisha Amari, Business Development specialist at AMSBIO said “iPSCs are increasingly being used for developing and discovering cellular medicines that have the potential to change the way doctors treat intractable diseases. She added “The GMP-compliant StemFit™ Basic04 CT is key in the manufacturing of iPSCs for use in cell therapy. Using StemFit™, we hope researchers can further advance translational research, drug discovery and development and help bring high-quality iPSC based- therapies from bench to bedside.”
For further information on StemFit™ Basic04 please visit https://www.amsbio.com/stemfit-stem-cell-culture/ or contact AMSBIO on +31-72-8080244 / +44-1235-828200 / +1-617-945-5033 / [email protected].
Founded in 1987, AMS Biotechnology (AMSBIO) is recognized today as a leading transatlantic company contributing to the acceleration of discovery through the provision of cutting-edge life science technology, products, and services for R&D in the medical, nutrition, cosmetics, and energy industries. AMSBIO has in-depth expertise in extracellular matrices to provide elegant solutions for studying cell motility, migration, invasion, and proliferation. This expertise in cell culture and the ECM allows AMSBIO to partner with clients in tailoring cell systems to enhance organoid and spheroid screening outcomes using a variety of 3D culture systems, including organ-on-a-chip microfluidics. For drug discovery research, AMSBIO offers assays, recombinant proteins, and cell lines. Drawing upon a huge and comprehensive biorepository, AMSBIO is widely recognized as a leading provider of high-quality tissue specimens (including custom procurement) from both human and animal tissues. The company provides unique clinical grade products for stem cells and cell therapy applications. This includes GMP cryopreservation technology, and high-quality solutions for viral delivery.







